Product News
View All Product News
Products
Please note : Any products described on this page are
for Research Use Only and not intended for clinical diagnostic procedures unless otherwise stated.
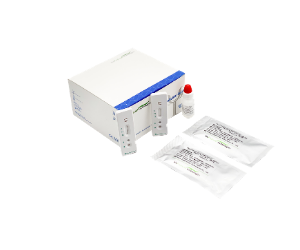
Novel Corona Virus (SARS-CoV-2) Ag Rapid Test
Clinical
Rapid chromatographic immunoassay (lateral-flow) for the qualitative detection of SARS-CoV-2 nucleocapsid antigens in nasopharyngeal swabs from individuals who are suspected of COVID-19 within the first seven days from the onset of symptoms.(Catalog Number: SC30107W)Nucleic Acid Extraction System SSNP-9600A
ClinicalLaboratory Equipment
SSNP-9600A Nucleic Acid Extraction System produced by Bioperfectus is a laboratory medical device, which integrates new technologies such as machine, electricity and computer software to enable automatic nucleic acid extraction of 96 samples maximumly in one time. Suitability: Match with virus nucleic acid extraction kit. High quality viral nucleicNucleic Acid Extraction System SSNP-3000A
ClinicalLaboratory Equipment
SSNP-3000A Nucleic Acid Extraction System produced by Bioperfectus is a laboratory medical device, which integrates new technologies such as machine, electricity and computer software to enable automatic nucleic acid extraction of 64 samples maximumly in one time. Suitability: Match with virus nucleic acid extraction kit. High quality viral nucleicNucleic Acid Extraction System SSNP-2000B
ClinicalLaboratory Equipment
SSNP-2000B Nucleic Acid Extraction System produced by Bioperfectus is a laboratory medical device, which integrates new technologies such as machine, electricity and computer software to enable automatic nucleic acid extraction of 32 samples maximumly at one time. Suitability: Match with virus nucleic acid extraction kit. High quality viralNucleic Acid Extraction Rapid Kit (Magnetic Bead Method)
ClinicalLaboratory Equipment
This kit uses magnetic beads and a buffer system with a unique separation function and is used in conjunction with a nucleic acid extractor to separate and purify high-quality viral nucleic acids from samples. The specially coated magnetic beads have a strong affinity for the nucleic acid in the sampleViral Nucleic Acid Extraction Kit (Magnetic Bead Method)
ClinicalLaboratory Equipment
This kit uses magnetic beads and a buffer system with a unique separation function and is used in conjunction with nucleic acid extractor to separate and purify high-quality viral nucleic acids from samples. The specially coated magnetic beads have a strong affinity for the nucleic acid in the sample underCOVID-19 Coronavirus Real Time PCR Kit
Clinical
COVID-19 Coronavirus Real-Time PCR Kit is an In Vitro Diagnostic (IVD) reagent applying fluorescent PCR technology and aiming to qualitatively detect Open Reading Frame gene region (ORF1a/b) and viral nucleocapsid region (N) of SARS-CoV-2 RNA from upper and lower respiratory tract specimens. Suitability: UpperNovel Corona Virus (SARS-CoV-2) IgM/IgG Rapid Test Kit
Clinical
The Novel Corona Virus (SARS-CoV-2) IgM/IgG Rapid Test Kit from Bioperfectus is an in vitro diagnostic test used for the detection of novel coronavirus (SARS-CoV-2) IgM and IgG antibodies. This product is based on immunochromatographic technology. The detection area of the strip has an IgM test line (T2 line),Influenza A and B Viruses Real-Time PCR
Clinical
Bioperfectus Technologies Influenza A And B Viruses Real-Time PCR Kit is an in vitro diagnostic test, based on real-time PCR technology, for the detection of Influenza A and B Viruses RNA. Samples can be obtained from throat swab, nasal and pharyngeal secretions and cell culture supernatant.COVID-19 Coronavirus and Influenza A/B Virus Real Time PCR Kit
Clinical
COVID-19 Coronavirus and Influenza A/B virus Real-Time PCR Kit are an In Vitro Diagnostic (IVD) reagent relying on fluorescent PCR technology for the qualitatively detection and differentiation of RNA from SARS-CoV-2, influenza A virus, and/or influenza B virus from upper and lower respiratory tract samples.Microbiology Product Areas
ClinicalNOTE: The products listed in this section may not be approved for clinical diagnostic use in your jurisdiction. You must assume these products are unregulated and therefore cannot be used for routine testing and reporting of results unless otherwise stated. | ||
| HPV | Show all suppliers | |
| Influenza | Show all suppliers | |
| Influenza H1N1 | Show all suppliers | |
| Mpox (monkeypox) Virus | Show all suppliers | |
| Respiratory Viruses | Show all suppliers | |
| SARS-CoV-2 | Show all suppliers | |
| Sexually Transmitted Diseases (STD/STI) | Show all suppliers | |
| Zika Virus | Show all suppliers | |