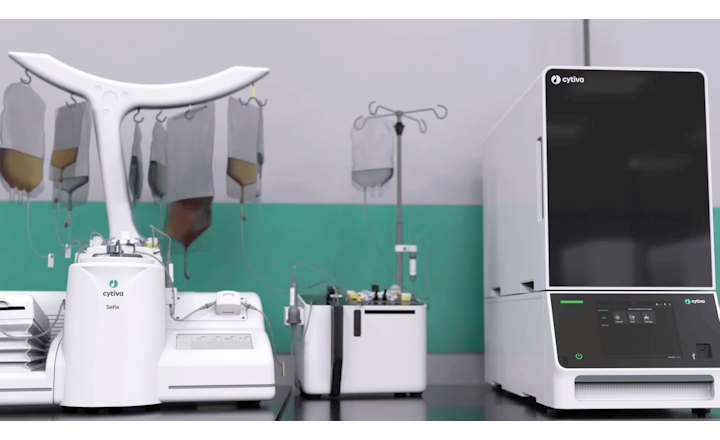
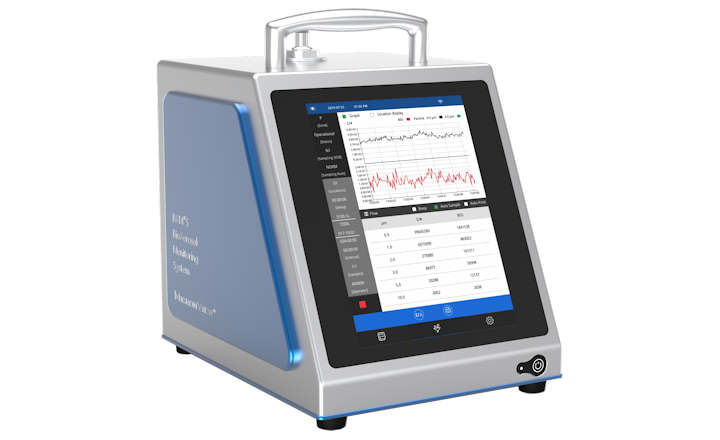
In the rapidly advancing field of cell and gene therapies, ensuring quality, safety, and efficacy in manufacturing is essential. Our cutting-edge technologies are designed to streamline production processes, enhancing quality control and providing the reliable testing systems that innovative therapies demand. In a landscape where time is critical—especially when patient outcomes are on the line—our solutions facilitate timely and precise results, empowering manufacturers to tackle challenges such as:
- Enhanced Quality Control: Robust testing systems that ensure compliance and safety.
- Improved Efficiency: Automated processes that reduce manual intervention and errors.
- Faster Time to Market: Accelerated workflows that help bring therapies to patients more quickly.
- Scalability: Flexible solutions that adapt to changing production needs and volumes.
Contamination Control Challenges Facing Cell Therapy Facilities
To explore these challenges in detail, access our webinar presentation highlighting the unique obstacles faced by cell therapy facilities and the need for tailored solutions. While some challenges are common across manufacturing sectors, their impact on cell therapy production is distinct. The webinar covers essential topics, including shelf-life considerations, contamination control, and cleanroom personnel management. Gain insights into risk reduction measures, achieving “right first-time” results, and the pros and cons of isolator technology. Join us as we navigate the complexities of cell therapy production and drive innovation in this critical field.
To access this informational webinar, visit our website.
There is no time to lose when a patient relies on your product. Our streamlined quality control testing solutions for cell and gene therapies yield safe, rapid, and accurate results.
Discover bioMérieux’s proven portfolio of value-added solutions.