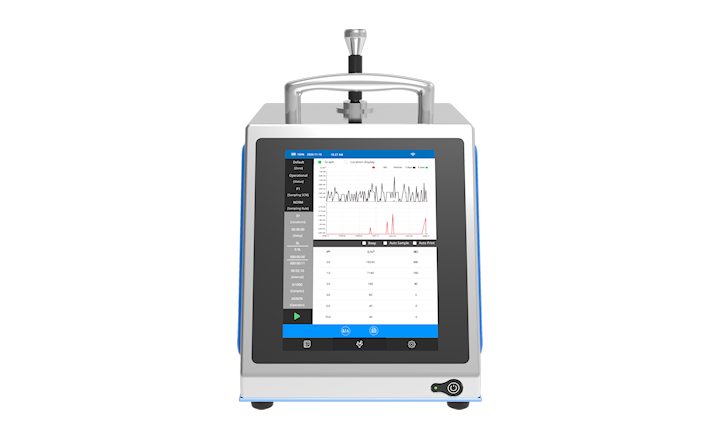
Lonza’s MODA-EM™ Version 3.3 software enables microbiology Quality Control (QC) laboratories to comply with new regulatory guidelines. QC facilities have to manage the daily challenges of QC monitoring while meeting changing regulatory compliance mandates. Lonza's latest software release meets these evolving challenges with additional features focusing on data integrity, data visibility and data review.
MODA™ Version 3.3 delivers improved workflow with better analytics and reporting than ever before. Complete with a streamlined implementation and a risk-based validation solution, this software is the paperless solution for QC laboratories.
“It is essential for QC laboratories to be able to readily adapt to changing regulatory guidance. To aid this compliance MODA™ Version 3.3 features a new, fully searchable audit trail that lets scientists easily track changes made through a sample’s lifecycle,” said Sinéad Cowman, EU Business Development Manager for Lonza Informatics. “The paperless efficiency features save time on many everyday activities, such as settling plate exposure times, organism identification and master data management and review.”
The new software has been designed to meet end user requirements and features enhanced scheduling and calendar capabilities for simplified sample management. It also offers web-based dashboards so that metrics and results can be accessed from anywhere with an internet connection. Scientists in QC laboratories are also supported with a validation documentation package that meets current industry standards and regulatory requirements, with execution and pre-validated service options available.
More information on MODA™ Version 3.3 and other QC testing solutions is available on www.lonza.com/moda